Empowered
by Movement
Our Vision
People with spinal cord injury will enjoy life in the ways that matter to them
Introducing ARCEX

Non-invasive spinal cord stimulation for people with spinal cord injury (SCI).
The ARCEX System is indicated to improve hand sensation and strength for people with a chronic cervical spinal cord injury.*
Now available for purchase.
Complete this form to learn more.
Visit the Center Locator to find
a center offering ARCEX near you
Discover our Therapy

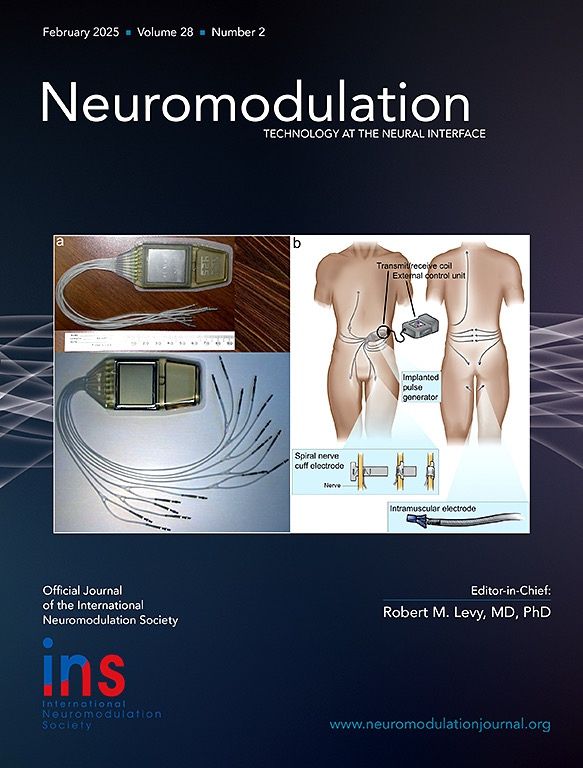
The ARCEX System is comprised of a Stimulator that delivers electrical pulses to the cervical spinal cord via Electrodes placed on the skin at the back of the neck.
ARCEX is meant to be used in conjunction with functional task practice in the clinic and with take-home exercises at home to support personalized rehabilitation in people with spinal cord injury.
Learn how to invest in ONWARD Medical
Meet our Team

We are a team of scientists, engineers, and physicians, collectively driven to create therapies that change lives.
We are driven to innovate, with 10 FDA Breakthrough Device Designation awards and nearly 400 issued or pending patents.
Up-LIFT pivotal trial results published in Nature Medicine

The Up-LIFT study was designed to evaluate the safety and effectiveness of our ARCEX Therapy to improve hand sensation and strength in people with chronic incomplete tetraplegia. The global study was conducted with 65 participants at 14 leading SCI centers in the US, Europe, and Canada.
Highlights** include:
-Primary safety and effectiveness endpoints met.
-90% of participants improved strength or function of their upper limbs.
-Upper extremity sensory scores improved significantly with device use.
-87% of participants reported improvement in quality of life.
-Improvement was demonstrated in participants up to 34 years post-injury.
Submit a Study Interest Form if you are interested in participating in our clinical trials
ARCEX is available
for purchase now
Read our Publications
View our Media Coverage
Explore our Blog
The ONWARD® Medical ARCEX® System is FDA cleared for use in the United States and CE marked in accordance with EU Medical Device Regulation 2017/745. ONWARD ARCIM® and ARCBCI™, alone or in combination with a brain-computer interface (BCI), are investigational and not available for commercial use.
*Indication for Use (US): The ARCEX® System is intended to deliver programmed transcutaneous electrical spinal cord stimulation in conjunction with functional task practice in the clinic and with take-home exercises in the home to improve hand sensation and strength in individuals between 18 and 75 years old that present with a chronic, non-progressive neurological deficit resulting from an incomplete spinal cord injury (C2-C8 inclusive). The ARC-EX System is intended to be operated in medical centers by Rehabilitation Professionals and at home by Patients and Persons Providing Assistance to the Patient as needed.
*Indication for Use (EU): The ARCEX® System is intended to deliver programmed, transcutaneous electrical spinal cord stimulation in conjunction with functional task practice in the clinic and with take-home exercises in the home to improve hand sensation and strength in individuals between 18 and 75 years old that present with a chronic, non-progressive neurological deficit resulting from an incomplete spinal cord injury (C2-C8 inclusive).
**Moritz, Chet, et al. “Non-invasive spinal cord stimulation for arm and hand function in chronic tetraplegia: a safety and efficacy trial.” Nature Medicine. 2024.